“This is the abstract of my Ph.D. thesis, as published by ACTA UNIVERSITATIS UPSALIENSIS, Uppsala Dissertations from the Faculty of Science and Technology (ISBN: 978-91-554-9650-0), entitled “Photonic crystals and photocatalysis. Study of titania inverse opals.”, published in 2016.
Due to an increase of human activity, an increase health risk has emerged from the presence of pollutants in the environment. In the transition to renewable and sustainable life style, treatment of pollutants could support the shifting societies. A motivation behind material research for environmental applications is to maximize the efficiency of the materials to alleviate environmental pollution.
In the case of titania, an increase of ultra-violet light absorption is needed to overcome its bandgap to produce reactive radicals, which is the basis for photocatalysis. It has been hypothesized that photonic crystal can enhance titania photocatalysis. They are structures made of at least two dielectrics with a high refractive index contrast, ordered in a periodic fashion. For a strong contrast, photonic band gaps emerge. The effect of the photonic band gap is to force complete reflection of the incoming light within its range and multiple internal reflections at its edges. By combining photonic and electronic band gap positions, it is possible to increase the absorption at the photonic band gap edges.
In this thesis, fabrication method and structural analysis of titania and alumina/titania photonic structures were presented. A thorough optical analysis was performed at all steps of fabrication – beyond what previously has been reported. The photocatalytic activity was measured with two setups. Fourier Transform Infrared spectroscopy combined with arc lamps and bandpass filters was used to monitor the degradation of stearic acid in ambient air. A home-built setup was used to degrade methylene blue in solution with ultra-violet illumination.
The results in this thesis show in general no correlation of the photocatalytic activity to the photonic band gap position, even though absorbance data displayed an increase absorption in this energy range. A more controlled environment might show the effect of the structure, as seen in some of the experiments.
An abstract is a structured summary, employed regularly for scientific publications, as a mean to sort quickly which article is interesting to read. The structure is usually as follow: a short introduction sentence, describing the major and keys elements (what is studied and why), few sentences on the method used (what did the authors did?) and the more important results (what was discovered that is new?), finished usually by a conclusive sentence (what should the reader remember most). The abstract for a book publication (which apply to a Ph.D. manuscript) is slightly more flexible and usually much longer. For my manuscript, I try to state first the problem, that is why we are doing the research in the laboratory, the second paragraph describes the material I use and why (theory oriented), followed by a third paragraph to explain in more details my experimental setup (what did I actually do?), the fourth and last paragraph summarize the main findings of my thesis (what did I discover?).
I. While the first paragraph is relatively easy to follow, the others need more explanations. Although, I would like to point out that pollutants originate from several different human activities and resides in both air and water. The obvious ones are the small particles and carbon-containing molecules in the atmosphere from the exhaust of petrol ignition (cars, boats, planes…). These cause respiratory problems and has been linked to a decrease in life expectancy. Several other less obvious sources of pollutants are, for instance, the emission from plastic-based furniture and from paints, varnish etc (Volatile Organic Compounds). Water can be polluted by the use of drugs (both for cattle and humans), by cleaning products, by wash out rain/snow from buildings, roads and such, and of course, the huge (seemingly insolvable) problem of tiny plastic particles. Clean drinkable water is still a luxe for the majority of the Earth population and is predicted to become a major source of conflict in the future (and is already starting).
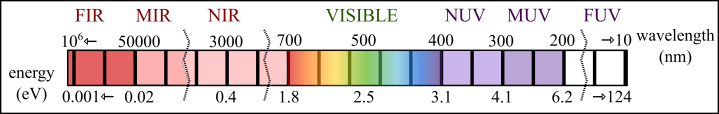
II. The second paragraph is dedicated to the theory behind the project, so it contains a lot of technical words, associated with air/water cleaning (for instance: “photocatalysis”), but also associated with physics concepts (“band gap”). I will try my best to use as little as possible of this special vocabulary in the following explanation. The first point to explain would be the expression “ultra-violet light absorption”: light can be described as both particle (called “photon”) and wave, which means that it can be described with its energy and its wavelength (distance between two maximum in a wave). Ultra-violet light originates naturally from our Sun (see figure 2) and is more energetic than the visible light (we do not see it, but other animals/insects can), as displayed in figure 1. It can also be produced by specific lamps (for instance Mercury based gas discharged lamp) for laboratory purposes. In science, we assign names to physical phenomena that happen to a light wave interacting with an object: the wave can be reflected (it goes back to the source), transmitted (it goes through the object) or absorbed (it is “consumed” by the object). There are variations of these, but these are the most important light-matter interaction. In the case of absorption, the wave (or photon) is losing its energy to the electrons in the material (electrons are building blocks of atoms, along with protons and neutrons). If the light is absorbed, it means that it is not propagating anymore (the wave stop).
Next is the word “titania”, which figures in the title of the thesis as well. This is a nickname for a compound called Titanium dioxide, with the chemical formula TiO2, which form naturally in an ore. It is composed of the atom of titanium (metal) and two atoms of oxygen, this is why it is called a “metal oxide”. It is an interesting material because it mixes some characteristics of metals (good at conducting electricity and heat) with that of semiconductors (in between complete insulators and metal). This brings us to the word “band gap”. It is a technical term to describe a phenomenon, which can be described theoretically but which is only a partial view of reality (the concepts are used in physics to help the mind understanding the physic). As such, it is a very loaded word, which one learns mostly at the university level, and in engineering and physics specialization at that. In chemistry, they have a similar concept but slightly differently defined, called “chemical potential”. In physics, it is linked to the electronic state of a solid: how do the electrons behave in the solid. Do they interact and “move” freely from atom to atom (metal-like)? Or are they attached (read attracted) to their atom (insulator-like)? If the electrons in the material move freely, there is no “band gap”, but if the electrons are restricted within the material, there is a “band gap”. What it reflects is that the nature of the electron energy (quantum) in a solid. If you take a simple atom of hydrogen, it has only a proton and an electron. But due to the small nature of these particles, they are not submitted to the everyday life physics we experiment, but to quantum physics. This “special” physics is fundamentally based on the uncertainty principle: if you know where you are in space, you have just a vague idea of your speed – and vice-versa. So, the electrons experience restrictions in space – as their velocity (momentum) is changing. This is reflected in the energy (potential energy, as in how much pull the nucleus of the atom has on the electron) it can take: the energy levels are discrete (it has to hop from ground energy – at rest – to an excited state – running – instantly). These discrete energy levels have a maximum filling capacity. For electrons, they have a very strict filling capacity – since electrons will repulse each other, as they have the same charged state – and because of their nature (spin 1/2). Due to this, the first energy level, the one closest to the nucleus in an atom, will only accept two electrons (so, in this case, we go from a hydrogen atom to helium atom). The next electron orbiting around the nucleus will not be able to stay the closest to the nucleus and will then occupy the next energy level (it will have more energy). There is an actual filling rule (following Pauli’s exclusion principle), defining the different energy levels – creating electronic orbitals: where electrons with the same energy have a probability of spatial orbit (on how it rotates around the nucleus). And this is expended to all atoms and molecules (accumulation of atoms). If you add more and more atoms together, in a repeated manner, to form a solid (an object we can manipulate), the original energy states of the electrons will have an influence on the degree of freedom the electrons experience in the final solid. And the “band gap” reflect the fact that no electrons from the solid can have a certain energy, they have to “hop” over a barrier to be excited. This is mostly valid for organized solids – with a repeated pattern and similar atoms. In the case of titania, there is a small band of energy states that are forbidden (that need to be overcome) and the electrons can only be excited (or be “running” instead of “resting”) if they receive an extra boost of energy. And this extra energy can come from light. With the right amount of energy (at least the size of the band gap), the electrons in the solid can absorb the photons traversing it.

Next, the text describes “photocatalysis” and “reactive radicals”. It ascribed to the description of chemical reactions on the surface of a solid. In a chemical reaction, molecules interact with each other to form other molecules, or a molecule separates into other molecules, sometimes realizing or consuming energy in the process. Photo originates from Greek and means light (so this is why the particle of light are called photons) and catalysis means to annul/to pick up. Put together, they describe the phenomenon of catalysis activated via light. Catalysis is a chemical phenomenon in which a catalyst will influence a chemical reaction, without changing itself, by reducing the amount of energy needed for a chemical reaction. In photocatalysis, the catalyst will absorb light and communicate this extra energy to promote a chemical reaction on its surface. What we called “reactive radicals” are molecules which are in a usually unstable because they have loosely attached electrons (called valence electrons) which would prefer to pair up with another electron (steal an electron from another molecule for instance) so that their total energy decreases. This is due to the electronic nature of the electrons (they are charged, negatively) and might go beyond the scope of this vulgarization. If you are interested in quantum physics (because this is basically it), I recommend to read or watch the Feynman courses. The thing to remember here is that the radical molecules want/need to obtain an extra electron. This means that they are extremely reactive, they will break chemical bonds to capture this extra electron. Their exact role differs from one chemical reaction to another, from one catalyst to another… and not all catalysis involve them. The most common ones are the oxygen and hydrogen radicals since they can originate from water molecules. Put all together: titania is a good photocatalyst because it can absorb ultra-violet light (from the Sun) and promote chemical reaction on its surface, by giving energy to the water on its surface, producing reactive radicals, which in turn will break other molecules (pollutants).

The following word “photonic crystal” then appear and it is proposed that theoretically (and with experimental evidence) using it would improve the photocatalytic activity of titania. So what is a photonic crystal? Well, from the “photo” part, it is clear that it deals with light and a crystal is a solid which possess a repeated pattern of atoms (for instance the diamond is a repeated structure of carbon atoms). So, it is a crystal or crystal-like structure which interacts specifically with light. More specifically, it is a technical term to describe natural structural colors. These can be seen on a large range of naturally formed structures: the most famous is the mineral opal (see figure 3). But it has been found on the skin of chameleons, of beetles, of fruits, on the wing of butterflies, on the feather of birds… Countless animals and insects display vibrant colors which originate from the interaction of their body with light. More precisely: the skin, feather, chitin… display extremely small features (in the nanometer range or 10–9 meter) that are repeated regularly at equally small intervals. It is the combination of repetition and small intervals that allow it to interact with light. As we have seen previously, the light can be described as a wave and therefore each wave will have a specific wavelength (the distance between two maximum). The wavelength of ultra-violet light is between 10 nm to 400 nm, while the visible light is defined by wavelengths of 400 nm to 800 nm. This means that structures with hundreds of nanometer repetition will interfere with the visible and ultra-violet light. You can see it as the difference it feels when you drive a car on a paved road compared to on a newly set asphalt: the asphalt also possess a structure, but it is too small to be felt, but the cobbles are much larger and can be felt. The structures observed on these natural examples will create a sorting of a sort, since it has repeated spots at a specific interval, some light with a specific wavelength will not be able to propagate (to go through the object): it will be reflected (in theory total reflection, in practice a small part can be transmitted). This is why these are called structural colors: certain wavelength of the incoming visible light are reflected back – so it is the observed color of the object. After this, we learn that this photonic crystal to enhance the photocatalytic activity of titania is formed by the use of two dielectric materials. So what are dielectric materials? It is a material that displays a slight separation of charges (positive vs negative) when under an electric field – for instance, under illumination. As a matter of fact, air is a dielectric material and so is titania. The abstract mentions “refractive index”. This is a constant that can be measured experimentally and predicted theoretically, unique for each material (even with the same type of atoms), defining at which velocity the light wave travel in the material, compared to the speed of light in vacuum (fixed and maximum, c). It is wavelength dependent (the light speed in the material will be different for different colors) and is often given at 500 nm (green) for material characterization. The refractive index of air is close to 1 (almost no difference compared to the vacuum light speed), while the refractive index of titania is around 3 (the light travel 3 times slower in the material than in vacuum). In the thesis abstract, the contrast between the refractive indices of the two material creating a photonic crystal is pointed as important for the emergence of a “photonic band gap”. Similarly to an electronic band gap where the electrons cannot exist in the material with a certain energy, a photonic band gap infer that photons cannot exist in the material with a certain energy. And theory shows (using Maxwell’s equations) that the stronger the refractive index contrast between the two building material, the wider the photonic band gap will be. It can be intuitively understood as follow: the light travels the fastest path, always, so it prefers to travel in a specific material, depending on its refractive index. By juxtaposing spatially and periodically two very different materials, it will split the light depending on its wavelength. And the wavelength in between these two extremes (the one that can travel in the low refractive index material, for us air, and the one traveling in the high refractive index material, here titania) will not find a preferential path, so will be reflected back. The stronger the interval, the more significant the effect. Note that the angle of incidence of light will also play a role here, since coming into a patterned surface at different angles will change the periodicity! This is why the wing of the butterfly Morpho Rhetenor change from dye midnight blue when observed from the side to light turquoise when observed from above. Interestingly, the light on the edge of the photonic band gap experienced a slower velocity than normal. Think of it as a gradual change of velocity imposed by the periodic structure. Since the visible/ultra-violet light is made of a wide range of different wavelengths, part of it will travel faster in the air around the periodic structure, another part will travel faster in the periodic structure itself, but the wavelength closest to the reflected one will be in the “in between”. It is the “edges” of the photonic band gap, and will travel in the different parts of the structure, but with an imposed periodicity, which does not “fit” as well than for the other wavelengths, therefore it will make them experiences in effect a slower propagation than in a normal continuous film of the same medium. It is often described as multiple internal reflections: the light with a specific wavelength preferentially traveling in one or the other material, but which cannot “fit” the periodicity, will “turn around” at the boundaries of the materials, several times before managing to “escape” it. But how such effect has an impact on the photocatalytic activity of titania? Well, first, it should be known that titania is not that good at absorbing ultra-violet light. It is due to the nature of its band gap, it is what we called an indirect band gap. This signifies that to be able to absorb the minimum amount of energy (the band gap energy) and be in an excited state, an electron needs the help of vibrations (collective vibrations in the solid are called phonons, and are treated as quasi-particle). Why is that translating to “bad” photons absorber? Well, if you have ever played (or seen) billiards, you know already how much more difficult it is to have three balls interact with each other, compared to two. It is exactly the same situation here: you need a photon, an electron and a phonon to all be at the right place, at the right time and at the right energy! All in all, titania can absorb ultra-violet light, but it is not super efficient at it. Another layer to the problem is added by the fact that the natural light emitted by our Sun is filtered by our atmosphere and a large portion of the ultra-violet light is already attenuated when reaching sea level (fortunately for life as we know it!). So why would we still persist in using titania then? Well, it has other advantages that are not to be easily disregarded: it is chemically inert itself (without ultra-violet light), it is abundant and cheap to extract from mined ores, it resists well to light degradation (since it absorb so little of the most energetic photons), it does not interact much with organic materials (titanium is often used in prosthesis placed inside the human body). This is where the idea that creating a photonic crystal with air and titania could help titania in absorbing more ultra-violet light: if you carefully craft the photonic crystal to have the ultra-violet light travel preferentially in the titania. Doing so, if you combined the edges of the photonic band gap (of the photonic crystal made of air and titania) to that of the electronic band gap (of titania), in theory you can “slow” down the ultra-violet light of the “right” energy (minimum energy gap in the electronic structure of titania – in other words, the minimum energy needed to excite an electron in the material) and therefore give more occasions for the photon absorption to occurs (you increase the probability of the photon-electron-phonon interaction). Doing so would increase the absorption efficiency, and therefore the energy transmitted to the surface of titania, where the chemical reaction happens. Well, that is all for the theory evoked in the abstract. What to retain?
(1) materials that can help chemical reactions just by being there exist (catalysts)
(2) catalysts that use light (photons) to promote chemical reactions exist too (photocatalysis)
(3) solids can have a special organization of their electrons (electronic structure)
(4) solids called semiconductors have a range of forbidden electronic states (band gap)
(5) the electrons in semiconductors have to obtain above band gap energy to be excited
(6) light (photons) can be absorbed by electrons
(7) light from the Sun can be visible, but also ultra-violet
(8) the wavelength of light is specific to its color (ultra-violet is 10 nm to 400 nm)
(9) titania has a band gap with energy close to the ultra-violet light
(10) titania is also a catalyst
(11) light velocity in a material is constant at specific wavelengths (refractive index)
(12) structural colors happen naturally: one color is reflected by a periodic structure
(13) existence of a photonic band gap: photons with specific wavelength are not allowed in the material (photonic crystal)
(14) the light can be manipulated in a photonic crystal by changing the refractive indices or/and the periodicity of the crystal
(15) light traveling in the periodic structure has to slow down (multiple reflections at borders)
(16) titania is not so good at absorbing ultra-violet light (indirect band gap)
(17) indirect band gap: interaction between electron, photon and solid vibration (low probability)
(18) ultra-violet light from the Sun is filtered by the atmosphere (low intensity at sea level)
(19) combining photonic and electronic band gap: increases the probability of ultra-violet light absorption in titania
(20) air/titania photonic crystal: increased photocatalytic activity
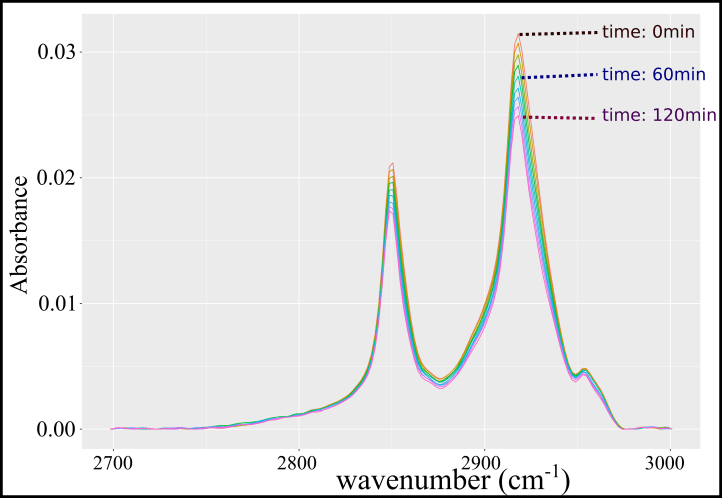
III. The third paragraph of this abstract describes the experimental procedures used, stating briefly that two different types of photonic crystals were fabricated: air/titania and air/alumina/titania. Alumina is an insulator, with a chemical formula Al2O3 (with a refractive index close to 2). An optical analysis was performed on the photonic crystal: this means that light was used to probe the samples. It is possible to measure reflected and transmitted light and calculate the absorbed part. This does not tell much on which type of measurements were used, but it can be suspected that both ultra-violet and visible light were used to measure the presence (or absence) of the photonic band gap of the different samples. Next, the abstract describes two setups/techniques used to measure the photocatalytic activity of the samples. The first is called “fast Fourier transform Infrared spectroscopy” (or FTIR). What is it? Well, infrared is light with wavelengths of 800 nm up to 1 mm (see figure 1)! This means that infrared light cannot be detected by the human eye (but can be by other animals) and that it has longer wavelengths than the visible light (so less energy). It is basically molecular vibrations – or, most simply, heat. Now, what is a Fourier transform? It is a mathematical operation. It deals with the transformation of an expression of a signal (be it light or sound…) in time/space to frequency (inverse of the wavelength). It is a very useful mathematical expression which allows the fast record of signals in time (all frequencies at the same time) and subsequently separating it (transform) to frequency-specifics (called a spectrum in science). So, in summary, the FTIR spectroscopy uses an infrared beam to probe molecules on a sample – to make them vibrates when the molecule absorbs it – and record the intensity change with time, but display the change by frequency. It is preferred this way because molecular bonds of specific molecules will have different specific frequency of vibration, allowing the monitoring of the molecular population on a sample surface. The abstract precise that “stearic acid” was used as an example of pollutant being degraded on the surface of titania. It is basically a component of fat and oils with a long chain of carbons (C18H36O2). The infrared light used in the FTIR setup makes the bond between the carbon and oxygen atoms vibrate (at a specific frequency) and this bond is broken with the decomposition of the molecule by the photocatalytic activity of the sample (so the sample need to be illuminated by ultra-violet light at the same time). The idea is to measure in time the disappearance of the vibrational signal of the molecule with its destruction, as seen in figure 4 (actual experimental result). The degradation rate is then calculated and represents the capacity of the sample to degrade molecules (the faster the better). The FTIR setup is set together with “arc lamps” (to provide an ultra-violet/visible light source) and “bandpass filters”, which are material designs to block and allow different parts of the light spectrum (it selectively let pass some wavelengths but absorb strongly the others). These filters can be used to change the type of light used in the experiments, without changing the light source (expensive and bulky).
The second experimental setup used to assess the photocatalytic activity of the sample is home-built: this means that the system was designed and produced by an individual and is not commercially available. Often, these system still are covered by a patent, but were not (at least yet) been bought by a company. This system was built by a fellow co-worker, Dr. Bozhidar Stephanov. It is composed of a chamber where the sample is set in water. A red laser is used to probe the water and a fluorescent ultra-violet tube is set above to illuminate the sample. The molecule degraded is “methylene blue“, which is a common dye (chemical formula: C16H18ClN3S). This means that its colors originates from the presence of a chromophore, where electrons are susceptible to be excited by visible light. In the solution, if orange light is absorbed, then, usually, the color perceived is the complementary color (blue). If the chromophore (the ring with sulfur) is destroyed or modified – as it is under ultra-violet light – the color of the solution with methylene blue shift from deep blue to transparent. And of course, using a red laser to pass through the solution will allow the possibility to correlate the degradation of methylene blue with the intensity of the laser: the less methylene blue, the stronger the laser signal after passing through the solution. This way, by calibrating laser intensity to known methylene blue concentration (how much of molecule per volume), it is possible to calculate the degradation rate. Figure 5 shows an actual measurement of the degradation of the dye during illumination.

So in this work, the photonic crystals were fabricated and optically probed to assess their photonic band gap (the light is strongly reflected at a specific wavelength, which can be easily predicted using Bragg-Snell‘s equation). To see if the photonic crystal structure has an effect on the photocatalytic activity of titania, the degradation rates were calculated from two different experiments: in air (using infrared light to probe a fatty acid) and in water (using red light to probe a dye).
IV. The last paragraph in the thesis drive the conclusion of all the experimental work: successful fabrication of photonic crystals but no correlation found between better photocatalytic activity and photonic crystal effect. Or rather, it is concluded that the plus given by the photonic and electronic band gap alignment is very small and negligible in contrast to other more important effects. In other words, the evaluation of the photonic crystal effect need to be made in a more controlled environment where all the other parameters are fixed (humidity, temperature, surface area, concentration of pollutant…).
